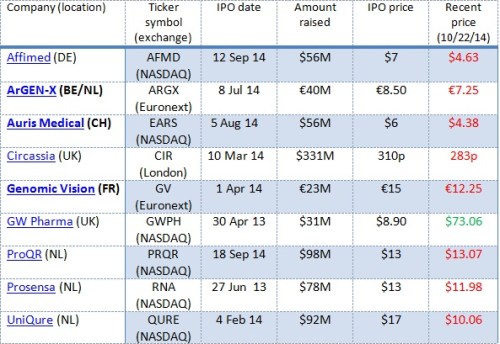
By Steve Dickman, CBT Advisors
More than ten years ago, I stopped using paper notebooks for my writing and consulting work. As someone who writes and thinks for a living, this was a big transition. But what a payoff I received in return! I no longer had to refer to handwritten notes or to type them later. My typed notes suddenly became searchable and editable. Since they are easy to access, they push me to new conclusions and new beginnings.
Just imagine how useful such a shift would be for biologists. Unlike the typical solitary writer or consultant, biologists work both on their own and collaboratively. Keeping their thoughts locked in paper notebooks has got slow down the free flow of ideas both between biologists as well as inside each one’s head. Indeed, putting biological data – in handwriting! – into a notebook that can only be read by one person seems almost criminal. “Cloud” software platforms have already enabled faster, more efficient collaboration in many industries and on many levels. Think Salesforce, Dropbox and Slack. Why not free the data and biologists’ early thoughts about it? Why not let the “hive mind” of the community go to work earlier and more efficiently? Over the internet, such sharing could break geographic boundaries and supercharge the thinking of biologists all over the world.
Especially in the areas of biology research that have a natural affinity for digital data and analysis – think genomics – biologists are already using online tools to record and share data. The same is true for chemistry, where protocols and starting materials (such as chemical precursors) are much more standardized. But in the less digitally aligned areas of biology, the shift from paper to electronic laboratory notebooks and similar online tools has been slow, sometimes glacially so. When it comes to their personal lives, the same biologists are emailing, texting and Slacking with the rest of us. But for many if not most biologists, when it comes to recording or sharing data, unless the lab procedure is performed by robots, the front end of the data collection process still looks like it did decades ago. Fresh data is recorded on paper or locked up within individual pieces of laboratory equipment. Then, later, perhaps, it gets transcribed into a sanitized version of biological reality.
The push for widespread electronic lab notebook (ELN) use is just beginning in biology. (The fact that these software tools are still even called “electronic lab notebooks” points to the fact that adoption has been repeatedly attempted – and has repeatedly failed – ever since “electronic” was the term for what we now call “online.”) One company I know performed an analysis that showed that electronic data recording and workflow management tools has only penetrated 8% of biology labs. Even if the actual number is larger – several industry-based biologists I asked said that 8% sounded low – the opportunity is undoubtedly huge. Consequently, a number of small and big software providers have plunged into this messy world, each hoping to convert biologists to a new paradigm or, better yet, to capture a mass movement that they believe is already underway. Some investors, including the Silicon Valley heavyweight firm Andreessen Horowitz, have announced a bold and public stake in the “clouding” of biology, as they call it, and promising big productivity and ease-of-use gains from that. The way that role model companies such as Salesforce and Dropbox have taken over other verticals, would certainly point to possible or even dramatic improvements. Seen in this light, the progress of the early entrants into ELN field would seem to be the leading indicators for when biology will shift more of its daily practice to the cloud and how completely and efficiently that can happen.
This piece aims to answer these key questions: Why has change been so slow? How is that starting to shift? And for what I believe to be at least a $10 billion question: will this transition happen quickly and powerfully enough to reward the companies, including those in the portfolios of investors like Andreessen Horowitz, currently hoping to capitalize on it?
Adoption is a tough slog
The challenges in converting biologists to cloud tools fall into a number of categories. To me, they break down like this:
Inertia and lack of immediate value: What has made Salesforce work in customer relations management, for example, is the obvious utility of the platform at local scale but especially globally. By contrast, at this early point in the ELN adoption curve, there is a lot of inertia retarding adoption and little history of productivity gains. One entrepreneurial molecular geneticist I know, currently working as a product development lead at a Bay Area molecular diagnostics company, said that he had thought about starting an ELN company back in 2012 but then abandoned it. Adoption of ELNs in biotech and academic biology labs “is very likely inevitable,” he wrote, “but the platform has to be heavily customized to each company’s unique needs, so it’ll likely be very complicated, need a LOT of effort to initiate, need extensive training for users to get it, require separate audits and so on.”
Even after biologists get over the initial activation energy barrier, the “aha” may not arrive immediately, if it does at all. One academic biologist I interviewed, Kristen DeAngelis, a junior faculty member at the University of Massachusetts in Amherst, put it like this: “When I was a postdoc at one of the government labs in 2007, there was a big push for electronic lab notebooks. They didn’t catch on. The software was clunky and slow, so it was not possible to capture observations as quickly as with writing; it was difficult to make sketches and record observations like numbers; and there is a big cost to switching, since lab notebooks have to stay in the lab for safety, so purchase of special tablets just for this was required and not many labs could make it work.” Set against these practical challenges, the promise of “big-data-like” returns on the initial ELN investment might be perceived as pie in the sky.
Secrecy and competition: Competition in academic biology, let alone in biotech, can sometimes be brutal. Every vendor makes it possible to limit outsiders’ access to online data but how many biologists will feel like they can trust this promise in light of the security breaches that have run rampant in, say, the financial sector? Especially because so many person-hours are invested in each hard-won experiment needed to win publication in a top journal, some academic biologists will likely prefer to go slow on uploading to any online platform including ELNs.
Degree of difficulty of biology: Some biological problems require inordinate amounts of faith and hard work, sometimes over years. In identifying new classes of receptor proteins (think about the netrins, for example, discovered by Marc Tessier-Lavigne, now president of Stanford University after three years of NOT discovering them) or puzzling out the intricacies of complex biological pathways, working solo or in a small, tight-knit group will be seen as an advantage. Easy connection with other biologists, not so much.
Lack of a common standardized computable biology language: This is a big one. Unlike, say, chemistry, in which most terms are unambiguous and new ones are rare, biology is a rapidly evolving field with little or no standardization of terms. Machine learning algorithms have been challenged by biology for a long time. As I wrote for the journal PLOS Biology in a different context fourteen years ago, ‘whereas extraction of person and place names from news text routinely reaches 93%, results in biology remain mired in the 75%–80% range.’ I quoted a brilliant structural linguist, Lynette Hirschman of the MITRE Corporation: ‘ “It’s a little depressing. Even something as simple as a slash may imply two different entities or a single compound.” Programmers eager to codify the rules of biology,’ my piece went on, ‘have been stymied by what one bioinformaticist calls “a sea of exceptions.”’ Even now, the lack of standard terms and the constant addition of new ones is a major hurdle for improving the utility of ELNs.
Both the software itself and the software-biologist interface is not doing the job: Working biologists from all parts of the spectrum – academia, the biotech industry and the pharmaceutical industry – reported major or minor difficulties with existing software packages. From pharma, where ELN use is typically mandatory, one senior neuroscientist I know reported that “The [ELN] software is actually a little slow. I believe the server is in France so it takes a few minutes to open the program and it is sluggish. That definitely aggravates people and makes them less inclined to adopt but honestly people don’t have a choice. It’s actually part of everyone in R&D’s performance review to adopt ELN best practices.” Adopt ELNs kicking and screaming! What a great marketing angle!
A very accomplished bioinformaticist responded to my email query about ELNs by saying that, barely one year into their transition to ELNs, his company had already split into two sets of users, one of which was continuing to use one platform while the other abandoned it and embarked on another. He wrote that the platform that part of the company abandoned – I won’t name it here – “…was advertised with the promise it can do anything — and that was the argument for buying it and [the accompanying] initial optimism. But that ‘do anything’ meant a lot of customization. Underneath it is an Oracle database that tries to be very, very generic. So you end up paying for [the vendor] to do that customization. So there was one [vendor representative] nearly living with us and still progress was very slow. That led to dissatisfaction.”
From academia, Megan Krench, who completed her neuroscience PhD in 2016 at MIT, reported that it was “astonishing” to her that academic biologists are not more avid users of ELN. She went on: “I’m not sure that we will see widespread adoption in the next ten years, since we haven’t seen it in the last ten. Everyone who has been a grad student in the last ten years was a digital native: why weren’t we all keeping ELNs?” This former student went on to say that “…of the roughly twenty labs I knew in grad school, only one had a lab-wide policy to use ELNs – and that was a young professors’ lab where he bought everyone iPads as a carrot to entice good bookkeeping. Of the roughly fifteen people in my lab, perhaps two of them kept an ELN instead a traditional paper one.”
DeAngelis, the University of Massachusetts biology professor, rounded out her comments by writing, “You didn’t ask [what my lab uses for a lab notebook]. I buy these by the case and issue them to all my lab members”:

The lab notebook of tomorrow? © 2017 TOPS Products (www.tops-products.com)
The lab notebook of tomorrow?
Attracting entrants
Despite or perhaps because of all these challenges, it seems like the ELN market, such as it is, has attracted more new entrants than ever. If anyone can foresee the drivers of change in the laboratory market, these companies can. In digging into this topic, Benchling is the company with which I spent the most time in researching this post (see disclosure). Benchling is in the portfolio of Andreessen Horowitz, which makes it one of the most high-profile players in a very diverse group of companies.
To read the rest of this post, click through to the original post here.